 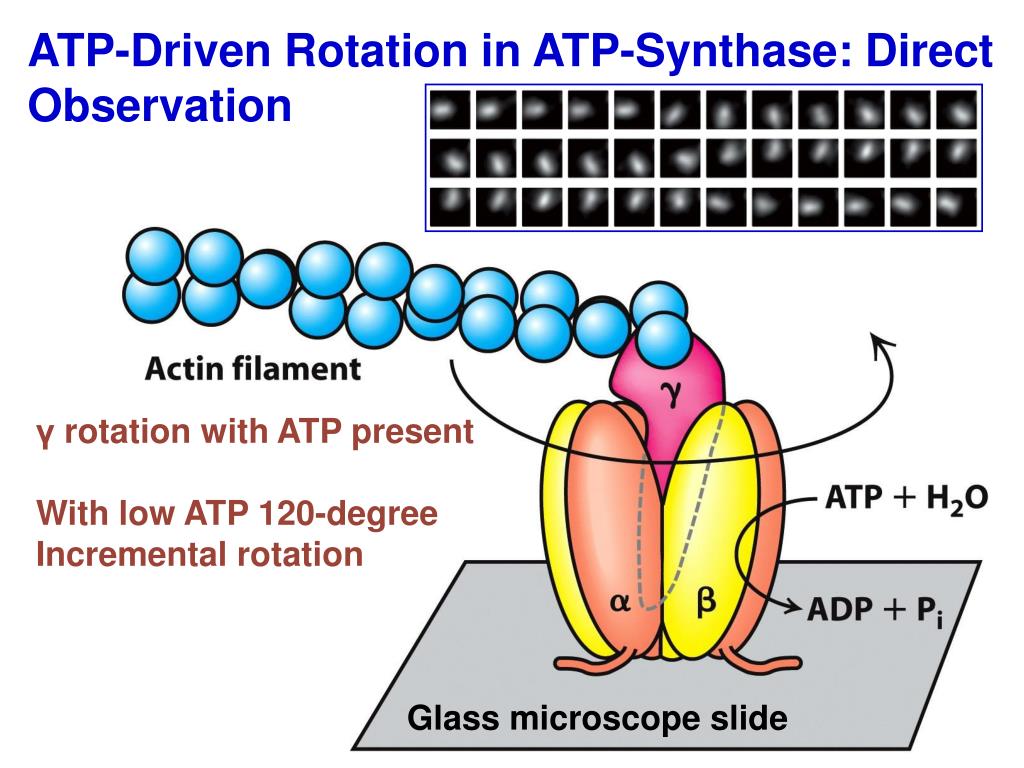
į O is a water insoluble protein with eight subunits and a transmembrane ring. Mitochondrial "delta" is bacterial/chloroplastic epsilon.Ĭalled "delta" in bacterial and chloroplastic versions.į O region F O subunit F6 from the peripheral stalk region of ATP synthase. These are particles of 9 nm diameter that pepper the inner mitochondrial membrane. The F 1 particle is large and can be seen in the transmission electron microscope by negative staining. The γ subunit allows β to go through conformational changes (i.e., closed, half open, and open states) that allow for ATP to be bound and released once synthesized. The other F 1 subunits γ, δ, and ε are a part of a rotational motor mechanism (rotor/axle). Three other subunits catalyze the ATP synthesis. Three of them are catalytically inactive and they bind ADP. Subunits α and β make a hexamer with 6 binding sites. The F 1 unit protrudes into the mitochondrial matrix space. The F 1 portion of ATP synthase is hydrophilic and responsible for hydrolyzing ATP. 
F O F 1 creates a pathway for protons movement across the membrane. F O on the other hand has mainly hydrophobic regions. F 1 has a water-soluble part that can hydrolyze ATP. F O causes rotation of F 1 and is made of c-ring and subunits a, two b, F6. Located within the thylakoid membrane and the inner mitochondrial membrane, ATP synthase consists of two regions F O and F 1. Subunits of the enzyme are labeled accordingly. Simplified model of F OF 1-ATPase alias ATP synthase of E. The F O, F 1, axle, and stator regions are color coded magenta, green, orange, and cyan respectively. Structure and function Bovine mitochondrial ATP synthase. This enzyme is used in synthesis of ATP through aerobic respiration. These functional regions consist of different protein subunits - refer to tables. The F 1 fraction derives its name from the term "Fraction 1" and F O (written as a subscript letter "o", not "zero") derives its name from being the binding fraction for oligomycin, a type of naturally derived antibiotic that is able to inhibit the F O unit of ATP synthase. An F-ATPase consists of two main subunits, F O and F 1, which has a rotational motor mechanism allowing for ATP production. This article deals mainly with this type. Organisms capable of photosynthesis also have ATP synthase across the thylakoid membrane, which in plants is located in the chloroplast and in cyanobacteria is located in the cytoplasm.Įukaryotic ATP synthases are F-ATPases, running "in reverse" for an ATPase. In prokaryotic cells ATP synthase lies across the plasma membrane, while in eukaryotic cells it lies across the inner mitochondrial membrane. This electrochemical gradient is generated by the electron transport chain and allows cells to store energy in ATP for later use. ADP + P i + 2H + out ⇌ ATP + H 2O + 2H + inĪTP synthase lies across a cellular membrane and forms an aperture that protons can cross from areas of high concentration to areas of low concentration, imparting energy for the synthesis of ATP.The overall reaction catalyzed by ATP synthase is: Stator is not shown here.ĪTP synthase is a protein that catalyzes the formation of the energy storage molecule adenosine triphosphate (ATP) using adenosine diphosphate (ADP) and inorganic phosphate (P i). Molecular model of ATP synthase determined by X-ray crystallography. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
